Reminder: Pharmboy is available to chat with Members, comments are found below each post.
Happy New Year PSW Members and Non-members alike! While in So. California the weather has not been so hot, the Pharma/Biotech sector was smokin' last year, with some big winners ( ) (CRIS (still holding after round one), ITMN, IMGN, ILMN, BIIB, BMRN, JAZZ, PODD, QCOR, ARIA (and here), ARRY, ARNA & DCTH (for some who got out early), VVUS (short)), some sad (
) (CRIS (still holding after round one), ITMN, IMGN, ILMN, BIIB, BMRN, JAZZ, PODD, QCOR, ARIA (and here), ARRY, ARNA & DCTH (for some who got out early), VVUS (short)), some sad ( ) losers (ARNA & DCTH for myself – although DCTH is back to positive finally, SPPI (for a small loss and then they took off – go figure), among others, and those that could still come in – PCYC, GXDX (mentioned in April, bought in December), BSDM (I guess the run up was baked in but still holding!) & TSRX.
) losers (ARNA & DCTH for myself – although DCTH is back to positive finally, SPPI (for a small loss and then they took off – go figure), among others, and those that could still come in – PCYC, GXDX (mentioned in April, bought in December), BSDM (I guess the run up was baked in but still holding!) & TSRX.
Many of our Big Pharma picks yielded some nice profits using the PSW strategy of How to Buy Stocks for a 15-20% Discount as they are right back where they started ( ) (MRK, LLY, GSK, BMY, NVS, SNY, PFE (PFE is higher as Phil had his buy writes, but for how long?), and FRX (short). So, if the stock is still a part of your virtual portfolio, sell the same ones from last year and enjoy those dividends and extra profits from the sale of calls and puts.
) (MRK, LLY, GSK, BMY, NVS, SNY, PFE (PFE is higher as Phil had his buy writes, but for how long?), and FRX (short). So, if the stock is still a part of your virtual portfolio, sell the same ones from last year and enjoy those dividends and extra profits from the sale of calls and puts.


Another year brings more FDA decisions (I will have a list for us to watch), something we try to avoid if at all possible due to the schizophrenic nature of the agency. I still like our IMGN, BIIB, ARIA, BSDM, GXDX and PCYC picks, with some more risky ones rounding out our biotech virtual portfolio (NNVC, NWBO, DSCO).
Up for today is Opexa Therapeutics (OPXA). OPXA is a publicly traded biotechnology company dedicated to the development of patient-specific cellular therapies for the treatment of autoimmune diseases. The company's leading product candidate, Tovaxin (under license from Baylor College of Medicine), has the potential to address the significant unmet medical needs of the large multiple sclerosis (MS) patient population.
Tovaxin is a personalized autologous immunotherapy that is not only manufactured for every individual patient but also is tailored to match each patient's evolving disease profile (much like Dendreon!). First, let's understand MS. MS is an inflammatory disease in which the fatty myelin sheaths around the axons of the brain and spinal cord are damaged, leading to demyelination and scarring as well as a broad spectrum of signs and symptoms. Disease onset usually occurs in young adults, and it is more common in females (Compston et al, 2008).
Figure 1. Nerve cell with myelin sheath (cells) around the axon. Figure 2. MS Degenerating the Spinal Chord.
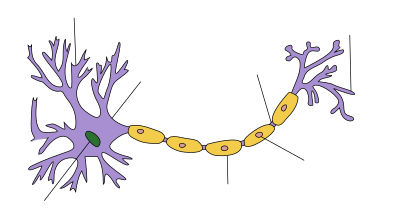

MS affects the ability of nerve cells in the brain and spinal cord to communicate with each other. Nerve cells communicate by sending electrical signals called action potentials down long fibers called axons, which are wrapped in an insulating substance called myelin. In MS, the body's own immune system attacks and damages the myelin. When myelin is lost, the axons can no longer effectively conduct signals. The name multiple sclerosis refers to scars (scleroses—better known as plaques or lesions) particularly in the white matter of the brain and spinal cord, which is mainly composed of myelin. Although much is known about the mechanisms involved in the disease process, the cause remains unknown. Theories include genetics or infections.
Now for the scientists at heart: Tovaxin consists of attenuated, patient-specific myelin reactive T-cells (MRTCs) against peptides of the three primary myelin proteins [Myelin basic protein (MBP), Myelin oligodendrocyte glycoprotein (MOG) and Proteolipid protein (PLP)] that have been implicated in T-cell pathogenesis of MS. (T-cells are one component of the immune system involved in fighting infection.) In preparing Tovaxin for a patient, the patient-specific MRTCs causing the disease are isolated from the blood and expanded in culture with specific peptides identified by assaying peripheral blood mononuclear cell (PBMC) reactivity against 109 peptides derived from MBP, MOG and PLP in the presence of antigen-presenting cells and growth factors. Myelin-peptide reactive T-cells are grown to therapeutic levels and cryopreserved. Prior to use, the MRTCs are expanded, formulated, and attenuated (by irradiation) to render them unable to replicate but viable for therapy. These attenuated T-cells are administered in a defined schedule of five subcutaneous injections. Patients are expected be treated with a new vaccine series (5 sc injections) each year based on their altered disease profile or epitope shift.
In 2008, topline data from the TERMS trial were presented at the World Congress on Treatment and Research in Multiple Sclerosis in Montreal, Canada, showing that the vaccine failed to meet its primary endpoint of reducing gadolinium-enhanced brain lesions. A non-significant trend towards a reduction in annualized relapse rate (ARR) compared with placebo was seen. The vaccine was safe and well tolerated. The most common side effect was mild injection site reaction, and no treatment-related side effects occurred. OPXA believed that the lack of significant efficacy could be due to the greater number of baseline brain lesions and lesion volumes in the drug-treatment group compared with the placebo group. At that time, a 1-year, open-label, extension study, OLETERMS, was underway. When this data started to surface in 2009, quality of life scores improved dramatically. Vision and activity demonstrated a significant improvement icompared with the placebo group. Patients in the Tovaxin group had also shown a 88% reduction in brain atrophy and a 59% reduction in absolute T-2 lesion volume. The US government is helping with the next phases of the companies studies, but they are not due out for a year or so. Interim results are due sometime this year. I recommend buying some stock here for any brief run up, and continue to accumulate on the dips.