Cliff Notes for “Swine flu: One Killer virus, three key questions.”
 By Ilene
By Ilene
Brendan Maher and Declan Butler, authors of a recently published Nature article “Swine flu: One killer virus, three key questions,” set out to answer three questions about the H1N1 virus. Here’s what they found.
How does it kill?
Sherif Zaki, head of the infectious-disease pathology branch at the Centers for Disease Control, discusses his team’s observations from their pathological studies.
1. The H1N1 virus penetrates deep into the alveoli (the terminal air sacs in the lungs). In contrast, the seasonal flu viruses tend to infect cells higher in the upper airways. This deep penetration is reminiscent of the action of the H5N1 avian flu virus. Zaki commented: “[H1N1] is like avian flu on steroids.” Why might this be?
Zaki says that his observations fit well with recent research looking at the mechanism of infection. A group led by Mikhail Matrosovich at Philipps University Marburg in Germany and Ten Feizi at Imperial College London studied sialyl glycans, glycoproteins that the flu virus binds to in order to gain entry to human cells. Although seasonal strains of H1N1 bind mostly to versions of the glycoproteins known as α2-6, the researchers found that the new pandemic H1N1 can also bind to a version called α2-3, which is found in greater proportion in the lower respiratory tract.
2. In patients who have died, co-infection was common with H1N1. Zaki’s group observed infection with bacteria such as Staph aureus or Streptococcus pneumoniae in about a third of the fatal swine-flu cases. However, in the remaining two thirds of fatal cases, the virus was lethal alone. The damage seen in the lungs is characterized as ‘diffuse alveolar damage,’ reflecting a very difficult to treat state of respiratory distress syndrome. According to Zaki’s research, about 90% of the fatal cases had some underlying medical condition.
Zaki expects the number of flu cases to increase as the flu season “ramps up.”
How does H1N1 spread?
Brendan Maher visited Peter Palese’s laboratory at Mount Sinai School of Medicine where testing for how the viruses spread is being carried out. Researchers John Steel and Anice Lowen compared the transmissibility of H1N1 to that of seasonal flu using a guinea pig model. Their data indicate that the H1N1 virus transmits as efficiently as seasonal flu viruses, which is consistent with real-world data showing rapid spread.
Prior exposure to H1N1 and H3N2 subtypes of seasonal flu seemed to limit the animals’ infection with the pandemic H1N1 strain, supporting the proposition that the seasonal flu vaccine provides some cross-protection against the swine flu.
What could it turn into?
Declan Butler spent time at the Jean Mérieux/INSERM biosecurity level four (BSL-4) facility in Lyon, France, where researchers are evaluating the likelihood that the pandemic H1N1 virus will ressort with the avian flu virus to create a deadly recombinant killer virus. In April, when the H1N1 pandemic began, priority lab time at the facility was allocated to Bruno Lina, a virologist at the CNRS, France’s basic-research agency, to conduct viral reassortment experiments. Although the pandemic H1N1 virus is not a BSL-4, the health ministry in France requires BSL-4 precautions for the reassortment experiments.
Butler writes:
The eight genes in the flu virus’s segmented genome are easily swapped between strains. But to be viable, new gene combinations must also be able to work together to package themselves into a virus particle. Viruses within a subtype tend to reassort with one another more easily and generate more viable reassortants than do those from different subtypes…
Lina is also preparing to submit a protocol to the facility’s scientific board seeking the green light to try to reassort pandemic H1N1 and the H5N1 avian flu virus. "It’s controversial research, but it is basic science that needs to be done," says Lina. H5N1 has killed more than half of the people it has infected since it resurged in 2003, but has rarely spread from one person to another. H1N1, on the other hand, seems to be as transmissible as seasonal flu, but mild in most people, if severe in some. The aim of Lina’s proposal is to find out the probability of a reassortant arising that combines the lethality of H5N1 with the transmissibility of H1N1…
Lina will use reverse genetics to generate a soup of reassortants, test whether any are viable, and if they are, assess their virulence and transmissibility. Because H1N1 and H5N1 are different subtypes, Lina does not expect them to swap genes easily. In 2005, he tried to reassort H5N1 with seasonal H1N1 and H3N2 viruses, without success. "After a year we only had three reassortants, and none was fit," recalls Lina, "they just don’t reassort well."…
Fortunately, Lina’s work suggests that viruses with the H1N1 subtype and avian flu viruses do not reassort easily into viable viral killing machines. However, the verdict awaits further research and real-life testing. Perhaps the most likely reassortment is between the pandemic H1N1 virus and the drug resistant seasonal H1N1 virus, resulting in the pandemic H1N1 strains showing increasing drug resistance.
Resistance can emerge by spontaneous mutation, but given that seasonal H1N1 is already resistant to the drug and spreads easily, reassortment is perhaps the most likely way that pandemic H1N1 will acquire resistance — especially as seasonal H1NI and pandemic H1N1 are the same subtype.
Tamiflu resistant strains have appeared in some countries but are not predominant. However, larger numbers of co-infections with the seasonal H1N1 circulating may result in more reassortments generating increased drug resistance as the flu season continues.

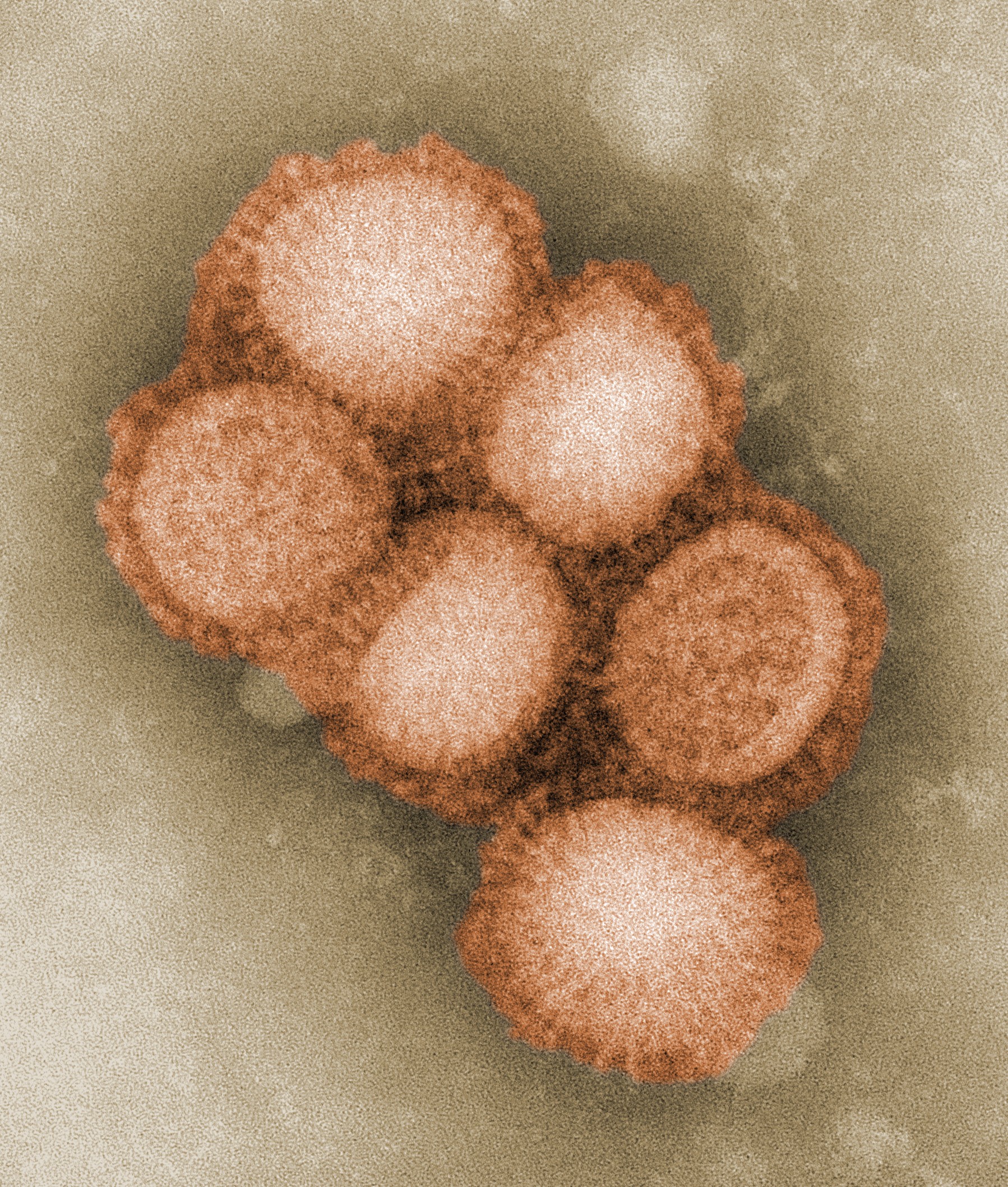
Photo: This colorized negative stained transmission electron micrograph (TEM) depicted some of the ultrastructural morphology of the A/CA/4/09 swine flu virus. Wikipedia.