Chocolate chemistry – a food scientist explains how the beloved treat gets its flavor, texture and tricky reputation as an ingredient

By Sheryl Barringer, The Ohio State University
Whether it is enjoyed as creamy milk chocolate truffles, baked in a devilishly dark chocolate cake or even poured as hot cocoa, Americans on average consume almost 20 pounds (9 kilograms) of chocolate in a year. People have been enjoying chocolate for at least 4,000 years, starting with Mesoamericans who brewed a drink from the seeds of cacao trees. In the 16th and 17th centuries, both the trees and the beverage spread across the world, and chocolate today is a trillion-dollar global industry.
As a food scientist, I’ve conducted research on the volatile molecules that make chocolate taste good. I also developed and taught a very popular college course on the science of chocolate. Here are the answers to some of the most frequent questions I hear about this unique and complex food.

How does chocolate get its characteristic flavor?
Chocolate starts out as a rather dull-tasting bean, packed into a pod that grows on a cacao tree. Developing the characteristic flavor of chocolate requires two key steps: fermentation and roasting.
Immediately after harvest, the beans are piled under leaves and left to ferment for several days. Bacteria create the chemicals, called precursors, needed for the next step: roasting.
The flavor you know as chocolate is formed during roasting by something chemists call the Maillard reaction. It requires two types of chemicals – sugar and protein – both of which are present in the fermented cacao beans. Roasting brings them together under high heat, which causes the sugar and protein to react and form that wonderful aroma.
Roasting is something of an art form. Different temperatures and times will produce different flavors. If you sample a few chocolate bars on the market, you will quickly realize that some companies roast at a much higher temperature than others. Lower temperatures maximize the floral and fruity notes, while higher temperatures create more caramel and coffee notes. Which is better is really a matter of personal preference.
Interestingly, the Maillard reaction is also what creates the flavor of freshly baked bread, roasted meat and coffee. The similarity between chocolate and coffee may seem fairly obvious, but bread and meat? The reason those foods all smell so different is that the flavor chemicals that get formed depend on the exact types of sugar and protein. Bread and chocolate contain different types, so even if you roasted them in exactly the same manner, you wouldn’t get the same flavor. This specificity is part of the reason it’s so hard to make a good artificial chocolate flavor.
How long can you store chocolate?
Once the beans are roasted, that wonderful aroma has been created. The longer you wait to consume it, the more of the volatile compounds responsible for the smell evaporate and the less flavor is left for you to enjoy. Generally you have about a year to eat milk chocolate and two years for dark chocolate. It’s not a good idea to store it in the refrigerator, because it picks up moisture and odors from the other things in there, but you can store it tightly sealed in the freezer.
What’s different about hot chocolate?
To make powdered hot chocolate, the beans are soaked in alkali to increase their pH before roasting. Raising the pH to be more basic helps make the powdered cocoa more soluble in water. But when the beans are at a higher pH during roasting, it changes the Maillard reaction so that different flavors are formed.
The flavor of hot chocolate is described by experts as a smooth and mellow flavor with earthy, woodsy notes, while regular chocolate flavor is sharp, with an almost citrus fruit finish.
What creates the texture of a chocolate bar?
Historically, chocolate was consumed as a drink because the ground beans are very gritty – far from the smooth, creamy texture people can create today.
After removing the shells and grinding the beans, modern chocolate makers add additional cocoa butter. Cocoa butter is the fat that occurs in the cacao beans. But there isn’t enough fat naturally in the beans to make a smooth texture, so chocolate makers add extra.

Next the cacao beans and cocoa butter undergo a process called conching. When the process was first invented, it took a team of horses a week walking in a circle, pulling a large grinding stone, to pulverize the particles small enough. Today machines can do this grinding and mixing in about eight hours. This process creates a smooth texture, and also drives off some of the undesirable odors.
Why is chocolate so difficult to cook with?
The chocolate you buy in a store has been tempered. Tempering is a process of heating up the chocolate to just the right temperature during production, before letting it cool to a solid. This step is necessary because of the fat.
Cocoa butter’s fat can naturally exist in six different crystal forms when it is a solid. Five of these are unstable and want to convert into the most stable, sixth form. Unfortunately, that sixth form is white in appearance, gritty in texture and is commonly called “bloom.” If you see a chocolate bar with white spots on it, it has bloomed, which means the fat has rearranged itself into that sixth crystal form. It is still edible but doesn’t taste as good.
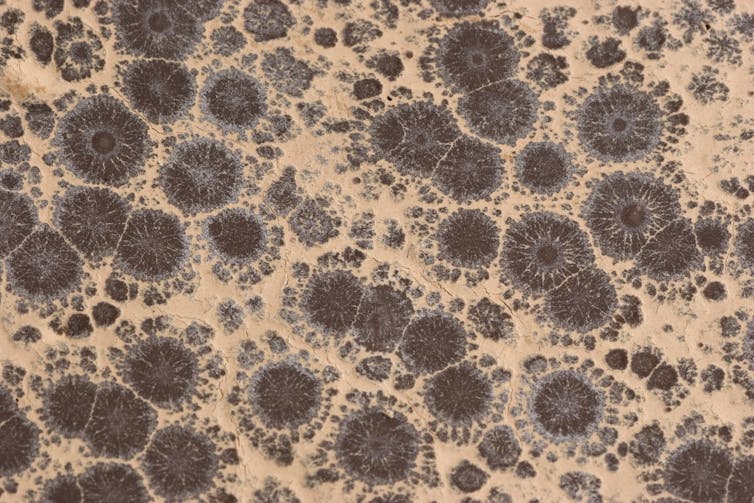
You can’t prevent bloom from happening, but you can slow it down by heating and cooling the chocolate through a series of temperature cycles. This process causes all the fat to crystallize into the second-most stable form. It takes a long time for this form to rearrange itself into the white, gritty sixth form.
When you melt chocolate at home, you break the temper. The day after you’ve created your confection, the chocolate usually blooms with an unattractive gray or white surface.
Is chocolate an aphrodisiac or antidepressant?
The short answer is, sorry, no. Eating chocolate may make you feel happier, but that’s because it tastes so good, not because it is chemically changing your brain.![]()
Sheryl Barringer, Professor of Food Science and Technology, The Ohio State University
This article is republished from The Conversation under a Creative Commons license. Read the original article.